

Product Name : Lorazepam
CAS : 846-49-1
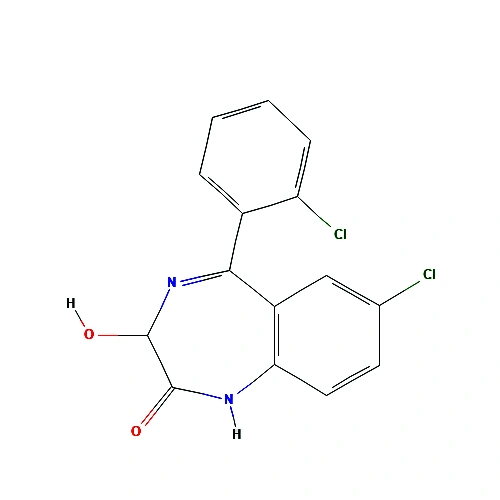
Molecular Formula : C15H10Cl2N2O2
Molecular Weight : 321.2 g/mol
Pharmaceutical Grade : BP / EP / IP / USP
Therapeutic Category : Antianxiety Agents, Anxiolytics, Anticonvulsant
Global calcium is one of the leading manufacturers and exporters of lorazepam CAS no. 846-49-1, API, (Active Pharmaceutical Ingredient). We support the customers with exhaustive documentation. As a USDMF & EU-GMP certified global company and an established hallmark for pharmaceutical standards, global calcium has stood the test of time since its inception in 1979.
As manufacturer of lorazepam we hereby state the following facts about the drug:
Lorazepam is a benzodiazepine with anxiolytic, anti-anxiety, anticonvulsant, anti-emetic, and sedative properties. Lorazepam enhances the effect of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) on the GABA receptors by binding to a site that is distinct from the GABA binding site in the central nervous system. This leads to an increase in chloride channel opening events, facilitation of chloride ion conductance, membrane hyperpolarization, and eventually inhibition of the transmission of nerve signals, thereby decreasing nervous excitation
Therapeutic uses and effectiveness:
Other related information:
Global calcium is a leading manufacturer of this drug. We manufacture this pharmaceutical drug and make it available to domestic and overseas market